
Global Medical Device UDI
All-in-one solution based on Track & Trace for global medical device UDI compliance
Improving the quality of medical device and increasing communication between MAH and end user are two common methods that MAH to choose to improving end user safety, which can be realized by medical device UDI (Meidical Device Unique Device Identification). To realize medical device physical secure on production, transportation, management, and safe to use, Track & Trace solution/solution based on unique code on batch level with DI (Device Identifier) is the way of implement global medical device UDI compliance. Furthermore, implementing PI (Production Identifier) on each unit (smallest sales unit) can provide more information about end user to form product and patient information integration. Combined JawaSoft's Track & Trace and NT-UDI is the solution for China medical device UDI compliance in one production line by code printing and aggregation on packaging. JawaSoft can provide all-in-one service in production which against lifescience threating; the production line upgrading or customized will improve competitiveness from brand reputation, production efficiency, future expansion and implement informatization under big data era.
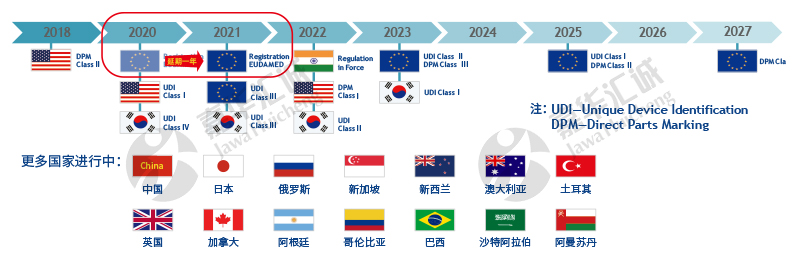
Track & Trace project in global medical device compliance
The categories of medical device products involve a wide range, and the packaging forms are diverse in specifications, and various types / languages of use method and introduction need to post on the package. Although the traceability regulations related to medical devices have begun in 1990s, the development on medical device growth too quickly to finalize the regulation in few years and implemente the regulation as pharmaceutical compliance. Therefore, the medical device regulations issued by various countries are gradually implemented according to the classification of medical devices.
Medical device UDI is the solution for:
• Driven by interests, counterfeit products on the market cannot be effectively verified in time, and inferior products cannot be accurately recalled and processed;
• In the case of rapid product updates or different production countries and sales countries, many products cannot be used accurately, resulting in medical accidents;
• When the feedback from the end user cannot be obtained, the product quality cannot be accurately improved, similar use errors cannot be avoided, and the required product type cannot be developed in time.
NT-UDI - YOUR GLOBAL MEDICAL DEVICE INFORMATION MANAGER
NT-UDI is a solution of UDI management for global medical device UDI information interaction.
JawaSoft patented platform (SaaS) NT-UDI could manage huge amount of data in easy way. NT-UDI could provide international UDI declaration and all-level of label design, and docking with production line (include coding unit, camera, labeller, etc.) with user friendly interface. Supporting all types of serialization and aggregation to improve production efficience.
Serialization & Aggregation - YOUR MEDICAL DEVICE QUALITY SAFEGUARD
Serialization & Aggregation software is a solution for anti-counterfeiting on each sales unit/batch.
Global medical device UDI compliance is aim to avoid wrong usage and prevent the unqualified product cannot recall on time caused life threatening. Many countries require serialization and aggregation for each batch-level of medical device to be traceable from manufacture to end user as regulation required.
Serial number (in form of barcode/Data Matrix/RFID) on each unit (e.g. item, bag, kit) package need be both AIDC and HRI, and it is unique in the world to be traceable basic information with 4W (what, when, where, why) . The TE Device to prevent illegal opening before it is opened by end user. Aggregation is assigned relationships between packaging levels (item/bag/kit, bundle, box, pallet), which from lower packaging level to higher level or through several packaging levels to ensure each item is tracable and the record about product can be traced for years.
Track & Trace - YOUR MEDICAL DEVICE LOCATION TRACKER
Track & Trace is a solution for batch-level of product traceability and anti-channeling.
The smallest sales unit is required to code print and inspect under high speed to qualify traceability requirement and high volume production requirement. For any type of complicated production line, JawaSoft can provide efficient solution on coding and inspection line of code printing, inspection, labeling, sealing whether semi-automatically or automatically under ISO standard.
Free consultation, CONTACT US!
若要访问和使用北京嘉华汇诚科技股份有限公司(以下简称"嘉华汇诚”)网站,您必须不加修改地完全接受本协议中所包含的条款、条件和嘉华汇诚网站即时刊登的通告,并且遵守有关互联网、万维网及或本网站的相关法律、规定与规则。一旦您访问、使用了嘉华汇诚网站,即表示您同意并接受了所有该等条款、条件及通告。
一、本网站上的信息
1、本网站上关于嘉华汇诚网站会员的注册信息均由会员自行提供,会员依法应对其提供的任何信息承担全部责任。嘉华汇诚网站对任何使用或提供本网站信息的商业活动及其风险不承担任何责任。
2、浏览者可以下载本网站上显示的资料,但不得用于任何商业用途,无论是否在资料上明示,所有此等资料都是受到版权法的法律保护。浏览者没有获得嘉华汇诚或各自的版权所有者明确的书面同意下,不得分发、修改、散布、再使用、再传递或使用本网站的内容用于任何公众商业用途。
二、版权和商标
2008-2013北京嘉华汇诚科技股份有限公司版权所有。所有的权利均在全世界范围内受到法律保护,除非有其他的标注或在此等条款和规则中被允许使用,本网站上可阅读和可见的所有资料都受到知识产权法的保护。
"jawasoft"、"jawasoft.com.cn"、"jawa-huicheng.com "、"jawa-huicheng.com.cn"、"jawa-huicheng. cn"、"jawa-huicheng.net"、"嘉华"、"北京嘉华"、"嘉华汇诚"和"嘉华汇诚LOGO” "嘉华软件"和"嘉华软件LOGO”商标或标识都是北京嘉华汇诚科技股份有限公司的商品及服务商标和商号。所有其他北京嘉华汇诚科技股份有限公司没有主张权利的商标和产品名称则可能是它们各自所有权人的商标或注册商标,未获得嘉华汇诚网或其他在本网站上有权使用商标的第三方的书面同意下,本网站不应理解为授权使用被展示于本网站的任何商标。
三、免责声明
嘉华汇诚在此特别声明对如下事宜不承担任何法律责任:
1、嘉华汇诚在此声明,对您使用网站、与本网站相关的任何内容、服务或其它链接至本网站的站点、内容均不作直接、间接、法定、约定的保证。
2、无论在任何原因下(包括但不限于疏忽原因),对您或任何人通过使用本网站上的信息或由本网站链接的信息,或其他与本网站链接的网站信息所导致的损失或损害(包括直接、间接、特别或后果性的损失或损害,例如收入或利润之损失,电脑系统之损坏或数据丢失等后果),责任均由使用者自行承担(包括但不限于疏忽责任)使用者对本网站的使用即表明同意承担浏览本网站的全部风险,嘉华汇诚、嘉华汇诚网站运营商和嘉华汇诚网站关联公司对使用者在本网站存取资料所导致的任何直接、相关的、后果性的、间接的或金钱上的损失不承担任何责任。